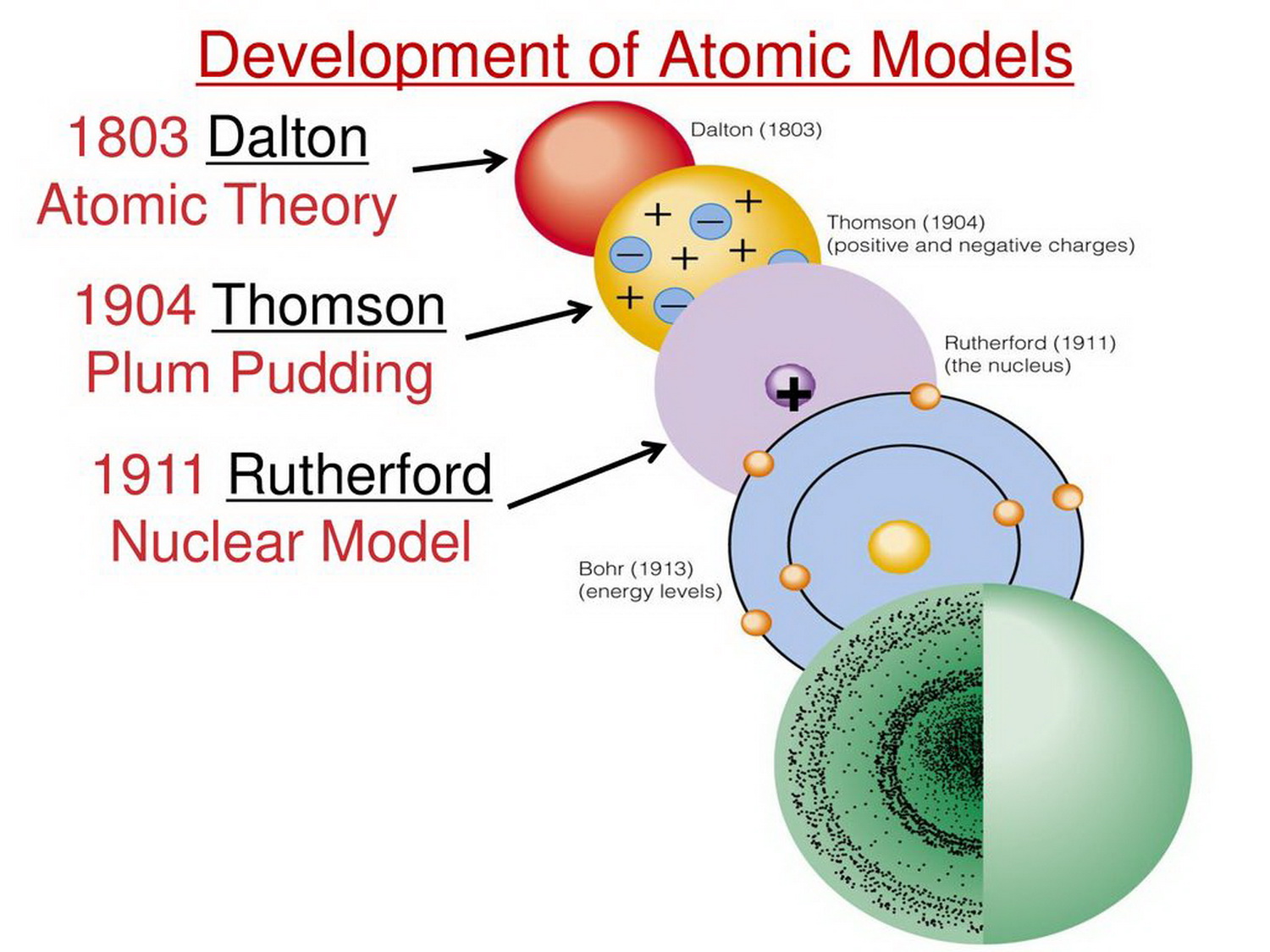
 Some particles even scattered back towards the source. Instead of virtually no deflection in all the alpha particles, while most particles passed through unaffected, some had huge deflection angles. If it is not true, there will be significant deflection If the hypothesis is true, there would be almost no deflection. The hypothesis is about deflection of particles, so the scientists measure deflection of particles. Scientists design an experiment which applies the hypothesis, where a measurable result will tell you if it is true or not.They predict a result that almost no deflection of alpha particles will take place, because if the observation was true then the fast-moving charged alpha particles will not be deflected by the sparse, ‘cloudy’ positive charge of the gold atoms. Scientists create a hypothesis or prediction to test the observation.Scientists take an observation from an existing topic: according to the plum pudding model, atoms are neutral overall, electrons are small negatively charged particles inside the atom so the rest of the atom must be sparse, dispersed cloud of positive charge. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
